Uses Of Diamond And Graphite Class 10

In this video of all about diamond in hindi we will learn diamond structure uses of.
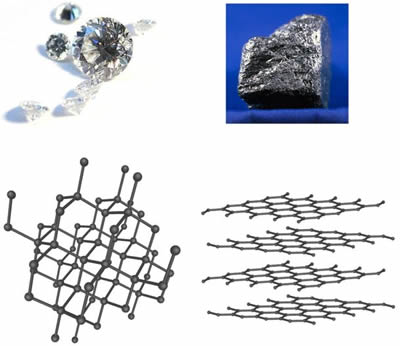
Uses of diamond and graphite class 10. In order to melt graphite it isn t enough to loosen one sheet from another. This is just a demo video for more videos and full syllabus please contact us on 8287971571 or 0261 4890014. Graphite ˈ ɡ r æ f aɪ t archaically referred to as plumbago is a crystalline form of the element carbon with its atoms arranged in a hexagonal structure it occurs naturally in this form and is the most stable form of carbon under standard conditions under high pressures and temperatures it converts to diamond graphite is used in pencils and lubricants. Structure of graphite and uses structure.
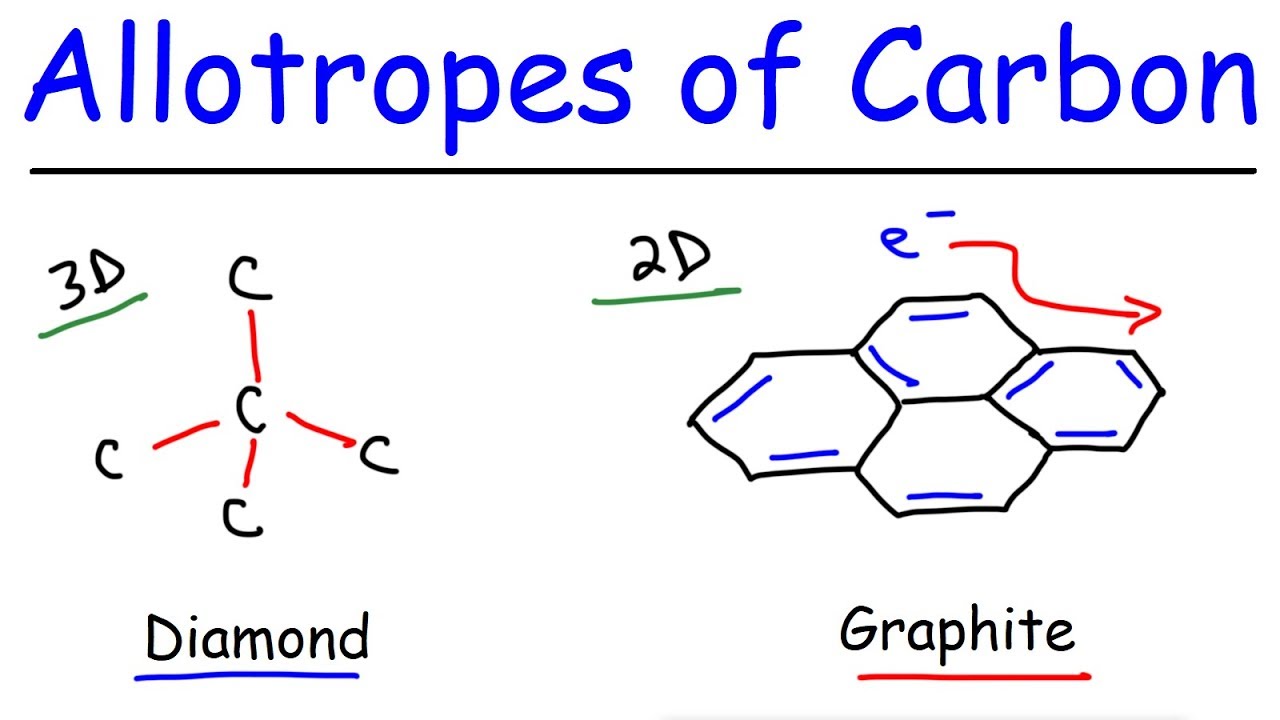
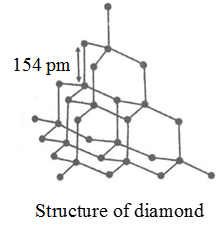
You have to break the covalent bonding throughout the whole structure. It is used in the making of jewelry. Graphite has a high melting point similar to that of diamond. Each carbon atom can form four covalent bonds.
This means that carbon atoms. Diamond dust is used to make saw blades and drill bits that can cut through just about anything. Graphene a naturally occurring ingredient in graphite has unique physical properties and is one of the strongest known substances. It is soft and slippery and its hardness is less than one on the mohs scale.
This derivative of graphite is further used in making lightweight and strong sports equipment. Graphite is mostly used for refractory battery steel expanded graphite brake linings foundry facings and lubricants. Graphite also has. 4 iupac group 14 of the periodic table.
Can form families of. These sheets are said to be 100 times stronger and 10 times lighter than steel. Unlike diamond graphite can be used as a lubricant or in pencils because the layers cleave readily. Diamond is used in the manufacture of filaments made of tungstens used for light bulbs.
Diamonds are used by most of the surgeons in the removal of the cataract from the eyes as a high precision instrument. Many are considering future applications in the field of medical and aerospace industry. Graphite is a stable form of naturally occurring carbon also known as plumbago blacklead or mineral carbon. It has a soft slippery feel and is used in pencils and as a dry lubricant for things like locks.
Graphite can be used to make graphene sheets. Carbon is an element.